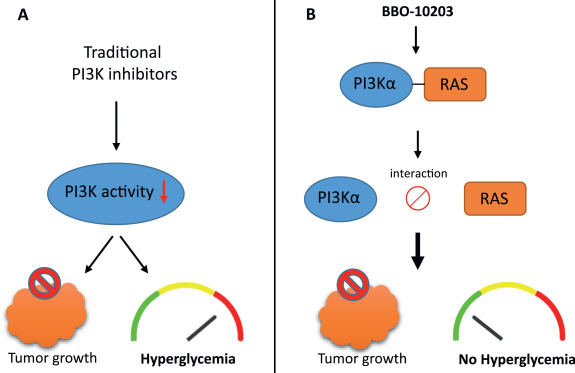
PI3Kα-RAS interface disruption as a new therapeutic strategy


The phosphoinositide 3-kinase (PI3K) family and the PI3K/AKT signaling pathway have been recognized as key players in oncogenic processes such as cell survival, metabolism, and metastasis. PI3K transduces signals from growth factors into intracellular responses by converting PI (4,5) P2 into PI (3,4,5) P3, activating the serine–threonine protein kinase AKT and downstream pathways. The PI3K/AKT pathway is tightly regulated, but in cancer, it can be constitutively activated through mutations in PI3K enzymes or receptor tyrosine kinases, such as EGFR or HER2. An alternative mechanism for activating the PI3K/AKT pathway involves the direct interaction between PI3K and the RAS protein. This interaction facilitates the recruitment of PI3K to the plasma membrane, promoting its catalytic activity.1 Disruption of the RAS-PI3Kα interaction alters tumour formation, highlighting its role in tumourigenesis.2 Based on the role of the PI3K/AKT pathway in cell growth and proliferation, it has been recognized as an attractive target for treating cancer. Several PI3K inhibitors have been characterized, some of which have already been approved for therapeutic use.1 However, most of these inhibitors are primarily ATP-competitive, which limits their specificity. While effective in suppressing tumor growth, their systemic application often causes hyperglycemia and insulin feedback by disrupting normal PI3K activity, limiting their tolerability and long-term clinical use3 (Fig. 1A). In a recent study, Simanshu DK et al4 presented an alternative approach by targeting the protein–protein interaction between RAS and the Ras-binding domain (RBD) of PI3Kα, thereby decoupling oncogenic activation from essential metabolic signaling.