Lactylation-driven METTL3 regulates wound healing by enhancing m6A/HNRNPA2B1/DNMT1 signaling in keratinocytes


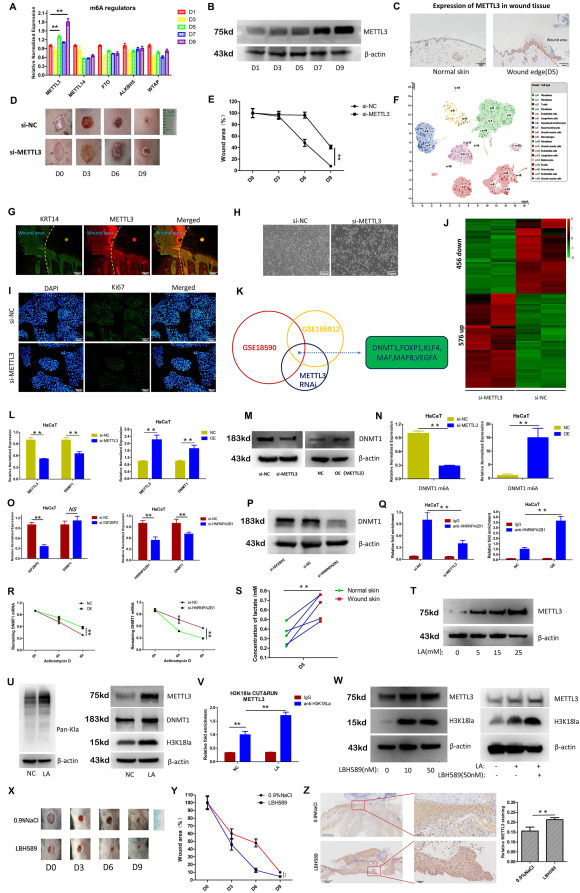
Chronic non-healing wounds, such as diabetic foot ulcers, represent a clinical challenge with an increasing incidence.1 A deeper understanding of the molecular mechanisms underlying wound healing is of great significance. RNA m6A modification is a widespread and important epigenetic regulatory mechanism,2 but its role in wound healing remains unclear. In this study, we found that METTL3, an RNA m6A methyltransferase, is a positive regulator of wound healing and that its low expression is closely related to chronic diabetic wounds. Mechanistically, we found that METTL3 regulates m6A modification of DNMT1 mRNA, increasing its expression and promoting keratinocyte proliferation. Additionally, we found that HNRNPA2B1 is the m6A “reader” that assists in regulating DNMT1 expression; HNRNPA2B1 can recognize m6A modifications on DNMT1 mRNA and increase its stability. Moreover, we discovered that lactate in the wound microenvironment accounts for the up-regulation of METTL3 expression during wound healing by inducing histone H3K18 lactylation. Finally, using a mouse wound model, we found that applying lactate or the lactylation “eraser” inhibitor LBH589 to the wound site promoted wound healing by up-regulating METTL3 expression. Our findings provide new targets for chronic non-healing wounds and suggest that lactylation modulation could serve as a potential therapeutic strategy for accelerating wound healing.