Macrophage κ-opioid receptor inhibits hypoxic pulmonary hypertension progression and right heart dysfunction via an SCD1-dependent anti-inflammatory response


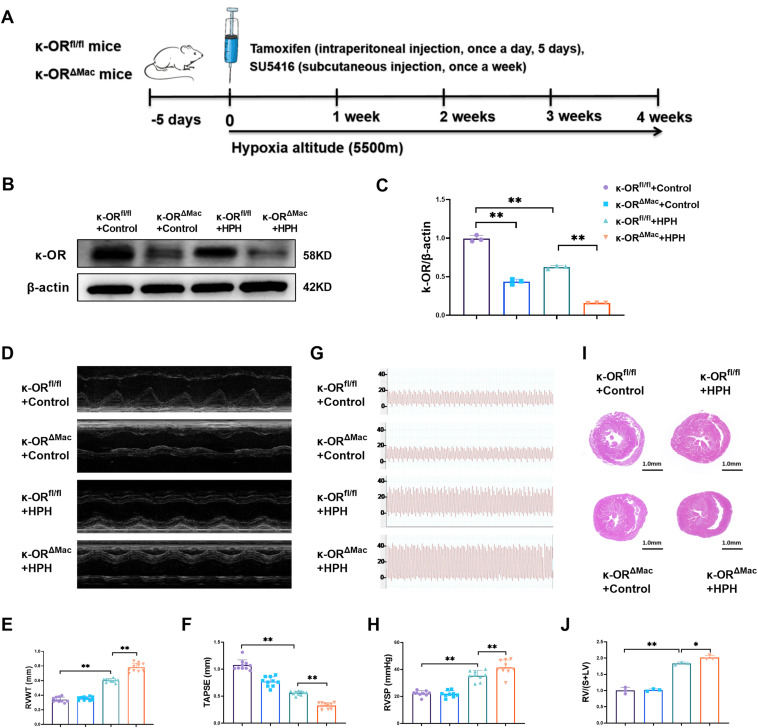
We aimed to investigate the effects and mechanism(s) of macrophage κ-opioid receptor (κ-OR) on macrophage inflammatory response and hypoxic pulmonary hypertension (HPH). Macrophage κ-OR-deficient mice (κ-ORΔMac) and their wild-type control mice (κ-ORfl/fl) were subjected to HPH or control groups. Mice with HPH presented significantly decreased expression of κ-OR in peritoneal macrophages. Compared with the κ-ORfl/fl + control group, the κ-ORfl/fl + HPH group presented increased right ventricular pressure, pulmonary vascular remodeling, and right ventricular hypertrophy and dysfunction; infiltration of M1 macrophages around pulmonary vessels; increased NLRP3 protein expression; and the release of the inflammatory cytokines. Macrophage κ-OR deficiency significantly aggravated the phenomenon mentioned above. At the cellular level, macrophages with κ-OR deficiency also aggravated lipopolysaccharide-induced inflammation. In addition, administering the κ-OR-selective agonist U50,488H significantly inhibited the inflammatory response in macrophages. The co-culture experiments revealed that U50,488H-treated macrophages inhibited the proliferation of pulmonary artery smooth muscle cells. Furthermore, our RNA sequencing and western blotting results revealed that κ-OR increases stearoyl coenzyme desaturase 1 (SCD1) expression in macrophages. Macrophage κ-OR knockdown significantly decreased SCD1 expression both in the lung tissues of HPH mice and in cultured macrophages. Moreover, SCD1 overexpression significantly suppressed the inflammatory response in lipopolysaccharide-treated macrophages, whereas the pharmacological inhibition of SCD1 increased the response. These results demonstrated that macrophage κ-OR inhibited HPH and right heart dysfunction by up-regulating SCD1, which inhibited macrophage inflammatory responses and pulmonary artery smooth muscle cell proliferation. This study provides more evidence to support the potential therapeutic role of κ-OR activation in the treatment of HPH.