m6A modification erased by ALKBH5 promotes tumor growth and metastasis via regulation of YAP/ZEB1 axis in NSCLC


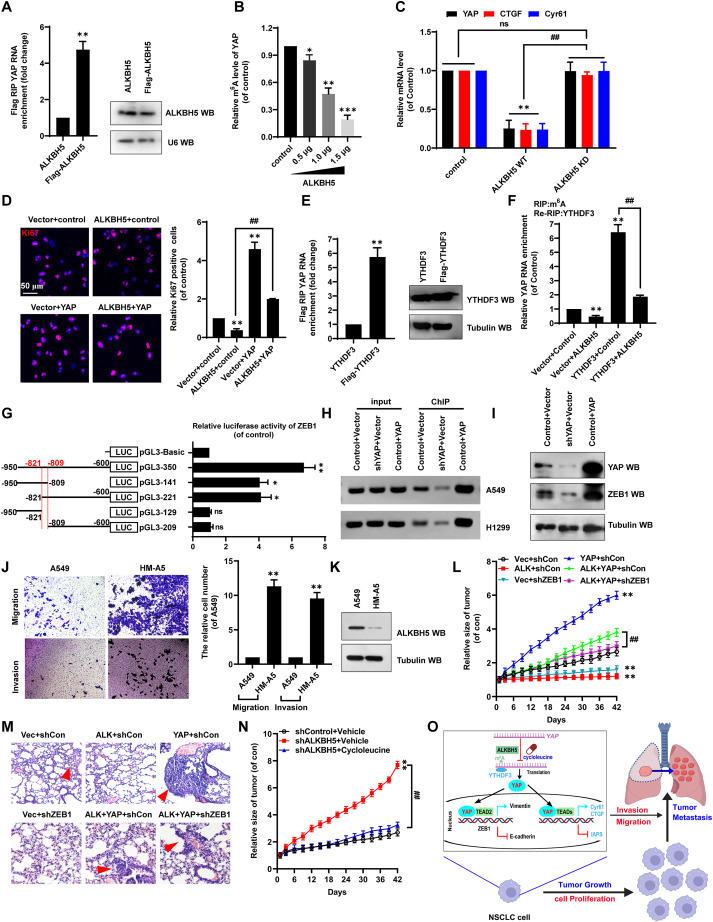
Early detection and effective treatment, as well as prevention of recurrence and metastasis, are crucial for patients with non-small cell lung cancer (NSCLC).1 Recent studies have shown that alkylation repair homolog 5 (ALKBH5) reverses m6A RNA methylation. Silencing ALKBH5 affects tumorigenesis and cancer progression under the action of m6A reading proteins, such as YTH domain family 3 (YTHDF3).2 The Yes-associated protein (YAP) pathway regulates cell proliferation, apoptosis, invasion, migration, and epithelial–mesenchymal transition (EMT),3 all processes that play a key role in tumor growth and metastasis.4 One notable EMT-related protein is zinc finger E-box–binding homeobox 1 (ZEB1), implicated in tumor progression.5 Currently, few studies have investigated the functions of these tumorigenic proteins in NSCLC. Here, our research revealed that ALKBH5, YTHDF3, YAP, and ZEB1 constitute the cellular axis regulating NSCLC cell proliferation, migration, invasion, and EMT in an m6A-dependent manner. Methylation inhibitor cycloleucine blocked this axis. Based on our findings, we propose that ALKBH5 plays an important supportive role in NSCLC tumor growth and metastasis. Thus, ALKBH5-mediated inhibition of YAP m6A modification is a promising novel target for NSCLC therapy.