A comprehensive analysis of immune checkpoint receptor–ligand pairs in aortic diseases highlights the immunosuppressive roles of CD155 and CD274


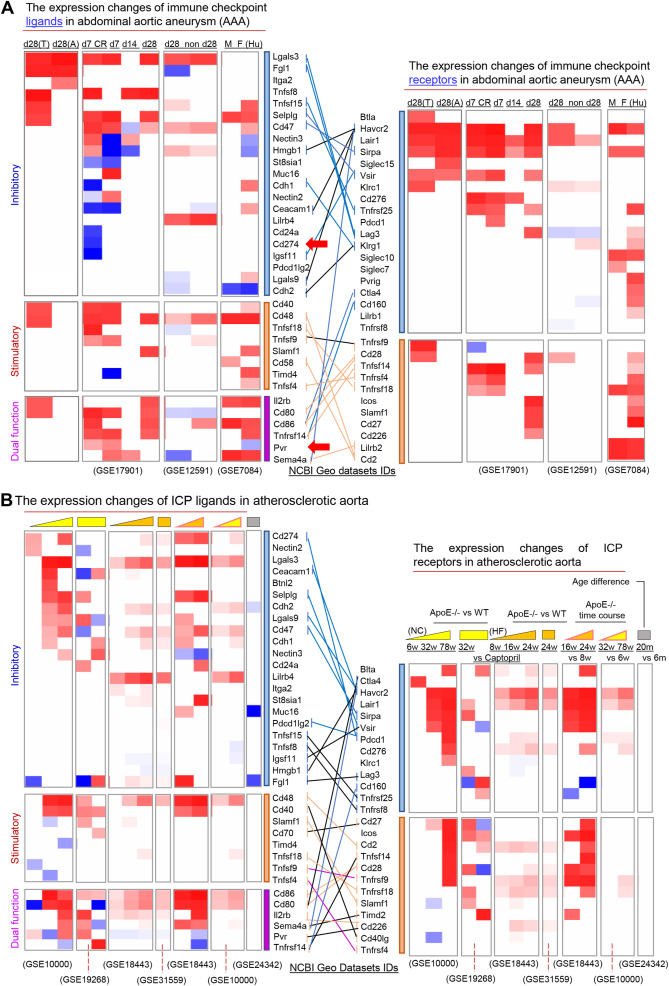
Immune checkpoints (ICPs) are immunologically functional membrane proteins essential for maintaining self-tolerance and regulating immune responses to prevent tissue damage.1 These molecules function through receptor–ligand interactions between T cells and antigen-presenting cells, balancing co-stimulatory and co-inhibitory signals. While therapies targeting ICP, such as inhibitors of programmed cell death 1 (PD-1)/programmed cell death ligand 1 (PD-L1) and cluster of differentiation 80 (CD80)/CD86/cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), have transformed cancer treatment, increasing evidence suggests that ICPs are also involved in the pathogenesis of cardiovascular diseases, including abdominal aortic aneurysm (AAA) and atherosclerosis.2 Notably, ICP receptors and ligands exhibit context-dependent roles in cardiovascular diseases, with diverse expression patterns and functions.3 Building upon our previous findings, identifying T cell-mediated vascular inflammation, including responses from CD4+ Foxp3+ regulatory T cells (Tregs), plays a critical role in AAA and atherosclerosis, we aimed to comprehensively characterize the ICP landscape in these conditions. To achieve this, we integrated Omics data with quantitative reverse transcription PCR validation to profile 58 receptor–ligand pairs (86 genes) across both murine and human datasets (Table S1). In our analysis of 10 AAA transcriptomic datasets (Fig. 1A), we observed consistent up-regulation of the inhibitory pairs CD47/signal regulatory protein alpha (Sirpa) and selectin P ligand (Selplg)/V-set immunoregulatory receptor (Vsir), as well as the stimulatory pair TNF superfamily member 14 (Tnfsf14)/TNF receptor superfamily member 14 (Tnfrsf14), particularly at early disease stages (Day 7 of angiotensin II infusion) compared with Day 28 in apolipoprotein E deficient (ApoE−/−) aortas. Notably, expression patterns were gender-specific; the CD47/Sirpa pair was up-regulated in females, while only Sirpa was increased in males. Selplg was up-regulated in both sexes, but Vsir remained unchanged, highlighting the complexity of immune modulation in AAA in a gender-dependent manner. These findings suggest that both co-stimulatory and co-inhibitory ICPs are inducible during AAA and may help maintain immune homeostasis and limit disease progression, especially during early stages.