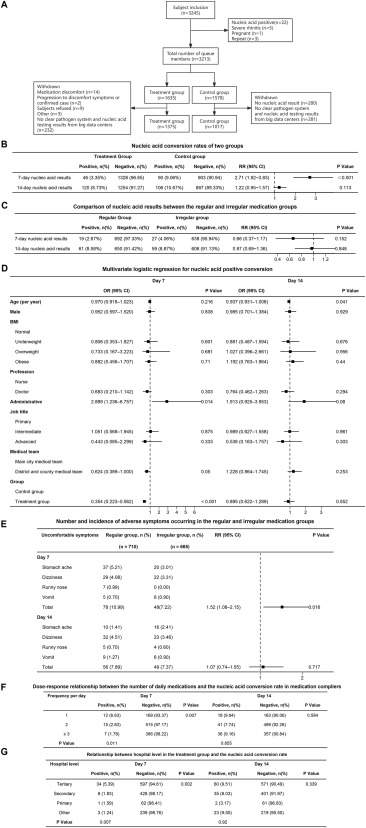
A cohort study on the A8G6 neutralizing antibody intranasal spray for preventing SARS-CoV-2 infection in healthcare workers


SARS-CoV-2 has evolved dramatically since the COVID-19 pandemic in Dec 2019, acquiring mutations that enhance transmissibility, infectivity, and immune evasion.1 Unlike other variants targeting lungs, Omicron altered entry pathways and promoted replication in the upper respiratory tract, resulting in more asymptomatic cases. This facilitated its spread and posed challenges to infection prevention.2 Healthcare workers face a higher risk of being infected and should seek to protect themselves from virus transmission while providing medical care for patients.3 Passive antibody therapy based on neutralizing antibodies has been found to protect susceptible populations with moderate to severe immunodeficiency or vaccine contraindications. Antibodies MY-586 and MY-558, obtained from recovered patients of SARS-CoV-2 infection, have shown strong neutralizing efficacy against various mutant strains of SARS-CoV-2.4 Preclinical toxicity studies in mice, monkeys, and healthy volunteers revealed that A8G6, the intranasal spray formulated with MY-558 and MY-586 at a mass ratio of 4:1, had high tolerance and no serious side effects. It has been confirmed to have potent efficacy in preventing SARS-CoV-2 infection in close contact with COVID-19 patients.5 Our study aimed to evaluate the efficacy and safety of A8G6 in safeguarding healthcare workers in field hospitals.