DNA damage-induced nuclear STING translocation orchestrates innate immune activation and chromatin remodeling


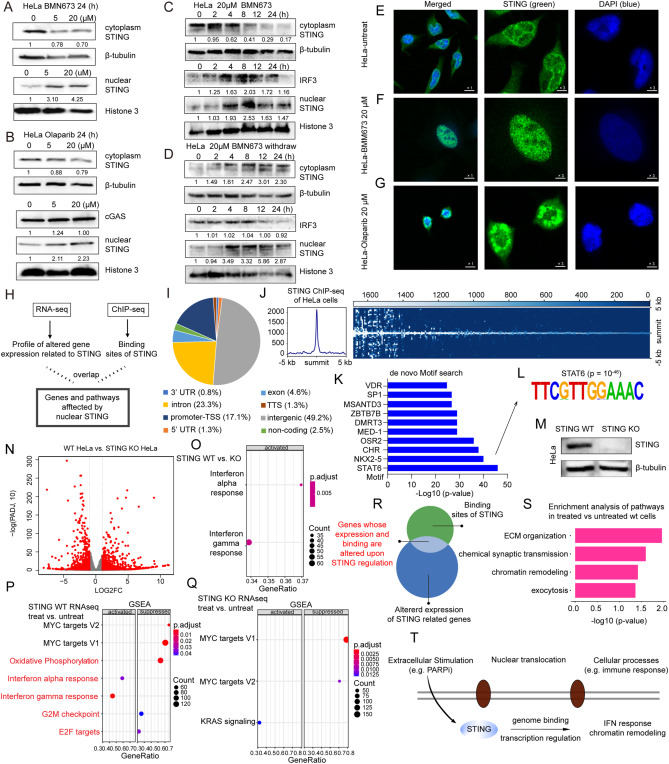
Stimulator of interferon gene (STING), a key innate immune adapter, senses cytosolic DNA to trigger type I interferon responses and is linked to cancer and autoimmune disorders.1,2 Beyond canonical signaling, STING acts as a proton channel promoting non-canonical autophagy and inflammation,3 while its nuclear translocation may facilitate DNA repair.4 Dixon et al identified STING redistributing from inner to outer nuclear membranes upon dsDNA or dsRNA stimulation.5 Nevertheless, the potential role of nuclear STING remains largely unexplored. In our study, we found that poly(ADP-ribose) polymerase (PARP) inhibitor-induced DNA damage triggered STING nuclear translocation in cancer cells. Chromatin immunoprecipitation sequencing (ChIP-seq) identified nuclear STING targets linked to chromatin remodeling and enriched transcriptional motifs. RNA sequencing (RNA-seq) data from STING wild-type and knockout cell lines confirmed nuclear STING's necessity for innate immune activation and chromatin remodeling. Our findings demonstrate that STING undergoes nuclear translocation, where it alters the immune response and chromatin remodeling of cancer cells. These results reveal novel molecular targets and non-canonical functions of nuclear STING, providing a framework for developing targeted cancer therapies to modulate STING activity.