Engineering HSV-1 for oncolytic therapy: From molecular entry mechanisms to retargeting strategies


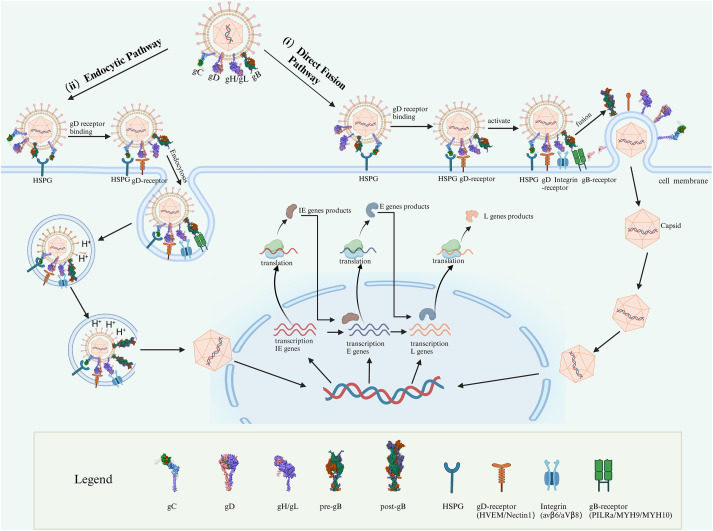
Oncolytic viruses (OVs) represent a cutting-edge approach to cancer immunotherapy, characterized by their ability to selectively infect and eliminate tumor cells while sparing healthy tissues. Among the diverse OVs, type 1 herpes simplex virus (HSV-1) stands out due to its robust oncolytic activity, genetic malleability, broad cell tropism, and well-documented clinical safety. Advances in genetic engineering have further amplified the therapeutic efficacy of HSV-1 by enhancing tumor specificity, replication efficiency, and immunogenicity. Clinically significant HSV-1-based oncolytic viruses, such as T-VEC and G47Δ, have gained regulatory approvals for treating melanoma and malignant glioma, respectively, highlighting their transformative potential in cancer therapy. The attenuation strategies employed in most oncolytic HSV-1 (oHSV-1) strains, while ensuring safety, often reduce viral replication and cytotoxicity. To address this limitation, retargeting strategies focusing on HSV-1 glycoproteins (gD, gH/gL, and gB) have been developed. These modifications aim to abolish canonical receptor interactions and achieve tumor-specific targeting through ligand-receptor binding. Recent breakthroughs in understanding HSV entry mechanisms have enabled the creation of fully retargeted HSV vectors with enhanced specificity and efficacy. This review explores the molecular mechanisms underlying HSV glycoprotein-mediated cell entry, examines recent advances in receptor-retargeted oHSV-1 engineering, and discusses the challenges and future directions in the development of oncolytic HSV-based therapies.