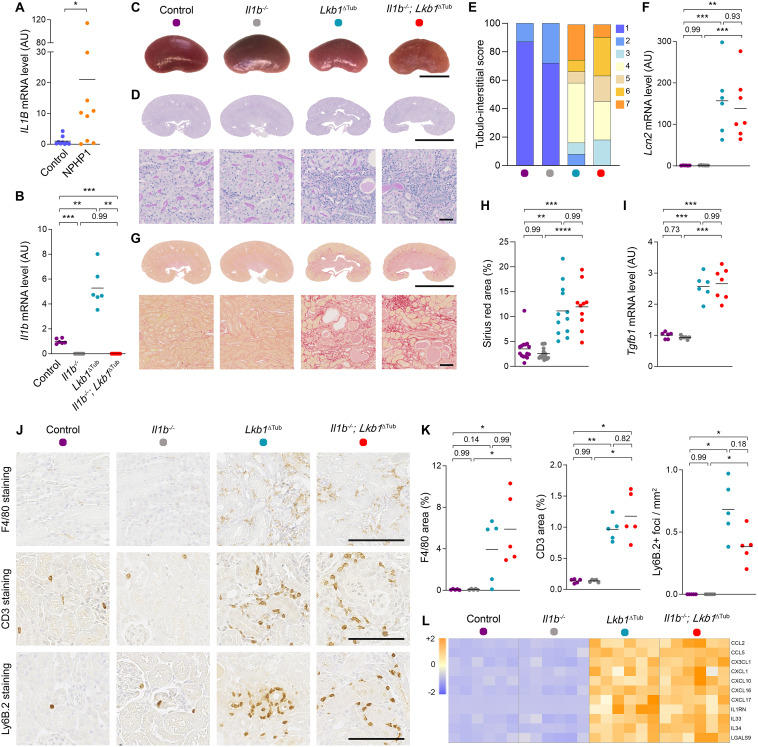
IL1β is induced in nephronophthisis but does not mediate kidney damage


Nephronophthisis (NPH), an autosomal recessive tubulo-interstitial nephropathy, is characterized by interstitial inflammation and progressive kidney fibrosis. To date, mutations in more than 25 NPHP genes have been associated with NPH, resulting in a wide genetic heterogeneity and overlapping clinical phenotypes. However, 53% of the patients with a genetic diagnosis have biallelic mutations in NPHP1.1 Fibrosis is caused by excessive matrix deposition, mainly by activated myofibroblasts. Inflammatory signals play a central role in the differentiation and expansion of myofibroblasts. The interleukin 1 (IL1) family of cytokines is one of the most potent triggers of innate immune response. The activity of IL1 is mediated by its type I receptor (IL1R) whose intracellular domain shares similarities with the Toll protein in fruit flies, known as the Toll interleukin-1 receptor (TIR) domain. When the cytokine binds, IL1R initiates a signaling cascade through recruiting cytoplasmic myeloid differentiation primary response protein 88 (MYD88), IL1R associated kinase 4 (IRAK4) and tumor necrosis factor receptor-associated factor 6 (TRAF6) resulting in the activation of the NFκB pathway, among others. In the kidney, in vitro studies reported that both IL1α and IL1β promote TGF-β production and fibronectin production in human proximal tubular cells. IL1β also promoted the conversion of kidney tubular epithelial cells into myofibroblasts, the primary drivers of collagen deposition.2 This finding was also supported by another study where treatment with IL1β triggered myofibroblast activation, matrix production, collagen deposition and fibrosis in kidney organoids.3