Nuclear and cytoplasmic USP30-AS1 coordinately regulate breast cancer progression through HnRNPF/p21 and EZH2/c-Myc/p21 axes


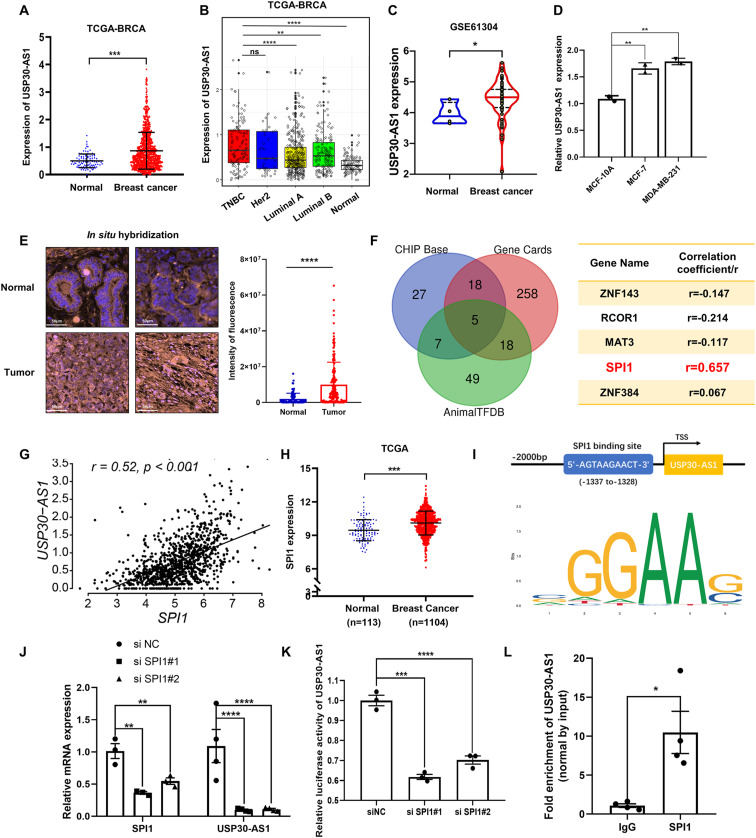
Emerging evidence suggests that aberrant expression of long non-coding RNAs (lncRNAs) is strongly associated with the occurrence and progression of breast cancer. Herein, we identified ubiquitin specific peptidase 30 antisense RNA 1 (USP30-AS1) as a markedly upregulated lncRNA in breast cancer tissues, and the transcription factor SPI1 functions upstream to regulate the expression of USP30-AS1. Gene set enrichment analysis suggests that USP30-AS1 may regulate cell proliferation. Knockdown of USP30-AS1 suppresses breast cancer cell proliferation and tumor growth by up-regulating CDKN1A/p21. Mechanistically, USP30-AS1 exhibits dual localization within breast cancer cells. In the cytoplasm, it interacts with HnRNPF, disrupting its binding to the p21 3′UTR, which destabilizes p21 mRNA and ultimately reduces p21 expression. In the nucleus, USP30-AS1 suppresses p21 transcription by enhancing the activity of c-Myc, a known transcriptional repressor of p21. USP30-AS1 binds to enhancer of zeste homolog 2 (EZH2), a histone methyltransferase, and prevents EZH2 from binding to the c-Myc promoter. This promotes epigenetic up-regulation of c-Myc by reducing H3K27 trimethylation. Together, these findings demonstrate the critical role of USP30-AS1 in breast cancer progression through HnRNPF/p21 and EZH2/c-Myc/p21 axes, highlighting its potential as a therapeutic target for breast cancer treatment.